What You Need to Know
The possibility of using stem cells to cure blindness inspires headlines and fills hearts with hope. For those living with vision loss from degenerative retinal diseases, it is crucial to understand what is fact and what is still a promise.
This article offers a sober and detailed overview of the advancements, focusing on the OpCT-001 cell therapy from BlueRock Therapeutics.
The core idea is revolutionary: growing photoreceptor cells (the cells that capture light) in a laboratory from induced pluripotent stem cells (iPSCs). These new cells are then injected under the patient’s retina to replace those destroyed by the disease. The ultimate goal is to restore the eye’s ability to “see.”
The first human clinical trial (Phase 1/2a), registered under the identifier NCT05908276, has been authorized and initiated 1. The primary focus of this initial phase is not to prove a cure, but to evaluate the safety of the procedure and the tolerability of the transplant.
Despite its enormous potential, the therapy faces significant challenges. The main one is the need for immunosuppressant drugs to prevent the patient’s body from rejecting the new cells. Furthermore, the technology has not yet been approved for commercial use, and its initial cost, when available, will likely be very high.
The key challenges of the therapy: the immunological barrier and the high financial cost. (Image credit: engeAI.com)
🧬 1. The Science: Rebuilding the Retina
OpCT-001 aims to treat diseases like retinitis pigmentosa, where genetic mutations cause the progressive death of photoreceptors (rods and cones). Without them, the retina loses its ability to convert light into neural signals, leading to blindness.
The BlueRock therapy works in three main steps:
- Creation: Adult cells are reprogrammed in a lab to become iPSCs.
- Differentiation: These iPSCs are carefully cultured to become photoreceptor precursor cells.
- Transplant: A suspension containing millions of these new cells is injected into the subretinal space so they can integrate with existing tissue 2.
The FDA’s granting of “Fast Track” designation in February 2024 acknowledges the seriousness of the condition and the therapy’s potential, but it is merely a procedural facilitator, not a seal of approval 3.
🔬 2. Clinical Trial Status and What to Expect
The Phase 1/2a clinical study is the first step in a long journey 1. Its objectives are pragmatic and methodical.
| Study Phase | Primary Objective | What is Observed |
|---|---|---|
| Phase 1/2a (Current) | Safety and Tolerability | Assessing whether the procedure causes inflammation, rejection, retinal detachment, or other serious adverse effects. |
| Phase 2 (Future) | Preliminary Efficacy & Dose | Checking for signs of improvement in visual function and determining the optimal cell dose. |
| Phase 3 (Future) | Efficacy Confirmation | Comparing the therapy against a control group in a large number of patients to prove clinical benefit. |
To date, no efficacy data has been published for OpCT-001 in humans. Any news of a “cure” is premature and speculative.
⚠️ 3. The Real Hurdles on the Path
The promise of cell therapy is immense, but the challenges are equally great.
Ongoing Immunosuppression: By using cells from a standard donor (allogeneic), the therapy requires the patient to take anti-rejection drugs. Although the eye enjoys a degree of “immune privilege,” it is not absolute 4. These drugs compromise the immune system and have their own side effects.
Cellular Integration: It is not enough to inject the cells; they must survive, organize themselves into the retina’s complex architecture, and form functional connections with existing neurons, a monumental challenge 2.
Industry Reality: Developing a therapy like this costs hundreds of millions of dollars. This financial pressure is reflected in corporate decisions. Between 2023 and 2024, BlueRock conducted layoffs and closed its R&D center in Cambridge, Massachusetts, to “prioritize the pipeline,” revealing the harsh reality of the industry 5.
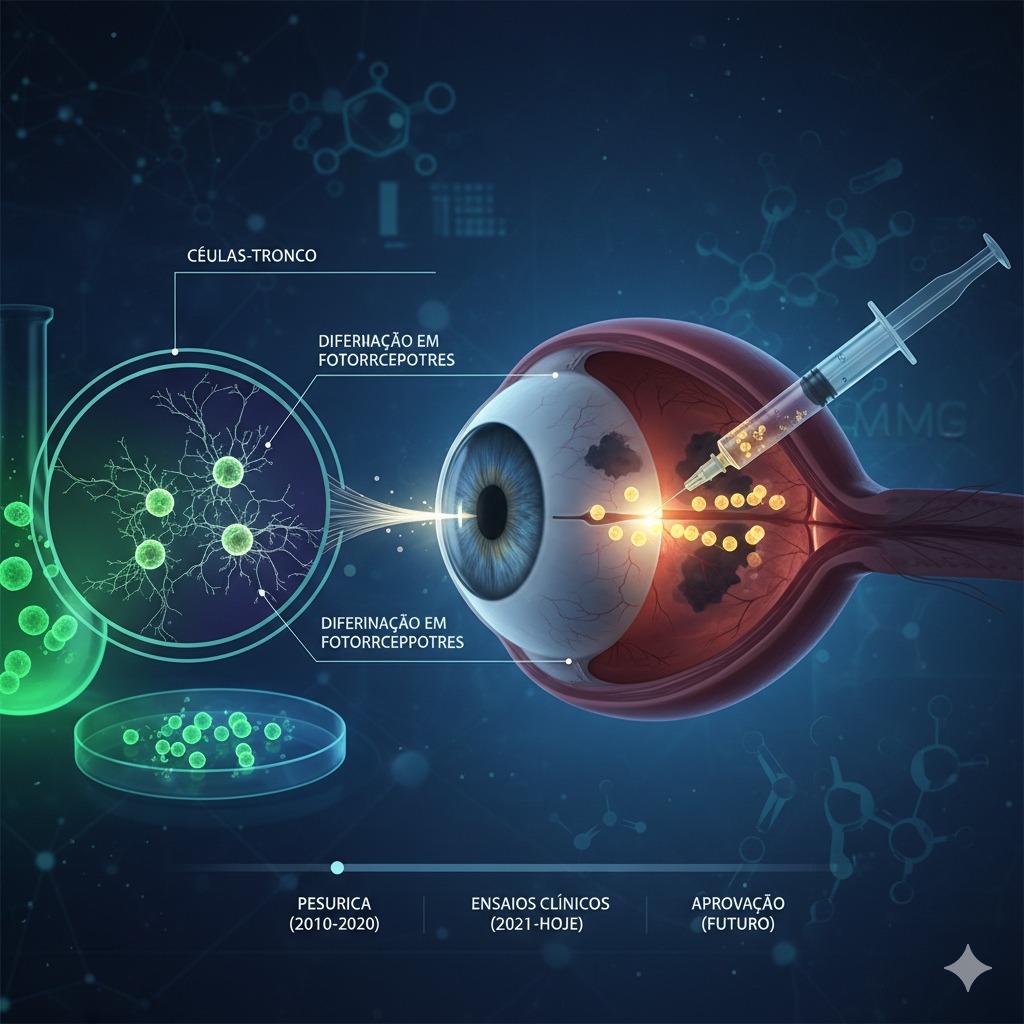
The OpCT-001 therapy process: from stem cell cultivation to their differentiation into photoreceptors and subretinal transplantation. (Image credit: engeAI.com)
🔮 4. The Future: A Sober Hope
OpCT-001 represents a genuine breakthrough. The possibility of replacing lost cells in the retina is, for the first time, a clinical reality under investigation.
However, the road to a functional, safe, and accessible cure is long. The ultimate goal for the entire field is to develop therapies that do not require immunosuppression, perhaps through gene editing 6. For patients and families, the message is one of cautious hope. Science is advancing, but it does so at its own pace—methodical and rigorous. An honest understanding of the process is what truly allows us to follow and support the advancement of science.
References
ClinicalTrials.gov. “A Study of OpCT-001 in Adult Participants With Primary Photoreceptor Diseases (STARGAZE).” Identifier NCT05908276. ↩︎ ↩︎
Mandai, M., et al. “Autologous Induced Pluripotent Stem-Cell–Derived Retinal Cells for Macular Degeneration.” New England Journal of Medicine, 2017. ↩︎ ↩︎
BlueRock Therapeutics. Announcements related to the “Fast Track” designation by the FDA, February 2024. ↩︎
Forrester, J.V., et al. “The eye’s immune privilege.” Trends in Immunology, 2008. ↩︎
FierceBiotech. News regarding the restructuring of BlueRock Therapeutics, 2023-2024. ↩︎
Cyranoski, D. “The CRISPR-edited immune-evading stem cells aiming for universal therapies.” Nature Biotechnology, 2024. ↩︎

