What You Need to Know
Type 1 Diabetes (T1D) is a chronic autoimmune condition that requires intensive daily management. This article provides a clear and realistic summary of the advancements in T1D treatment, with a focus on the promising VX-880 therapy by Vertex Pharmaceuticals.
The technology involves the transplantation of human beta cells derived from laboratory-grown stem cells. Infused into the liver, these cells restore the body’s ability to produce insulin autonomously. In trials, multiple volunteers have achieved insulin independence for over a year. However, the therapy is not yet approved and requires the use of immunosuppressive drugs. The initial cost, when available, will be high.
📜 A Century of Progress
T1D affects over 8 million people worldwide. Even with advanced systems, about 75% of patients do not reach the hemoglobin A1c target (< 7%). The century-long quest to eliminate insulin injections has now entered the era of cell therapies.
🧬 1. Brief History of T1D Treatments
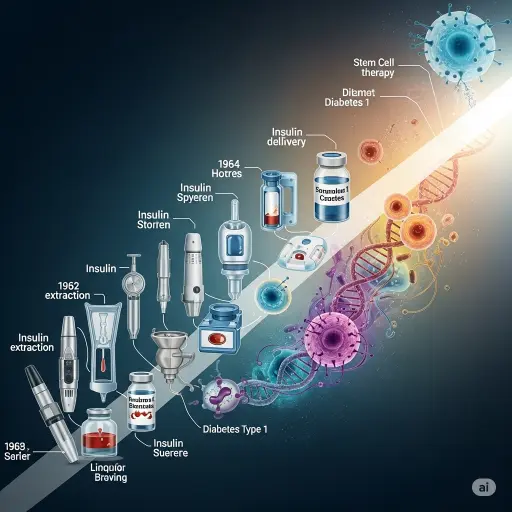
| Year | Milestone | Details |
|---|---|---|
| 1921 | Discovery of Insulin | Banting and Best reverse hyperglycemia in dogs. |
| 1922–70s | Animal Insulin | Commercial production from pig and cow pancreases. |
| 1982 | Recombinant Humulin® | First human insulin produced by genetic engineering. |
| 1990s–2010s | Rapid and Basal Analogs | Finer control of insulin action. |
| 2013–Today | Closed-Loop Systems | Integration of pumps and CGMs (“Artificial Pancreas”). |
| 2021–Today | Stem Cell Therapy | Clinical trials with Zimislecel (VX-880). |
🔬 2. Vertex’s Stem Cell Therapy: A New Era
Zimislecel (VX-880)
Vertex Pharmaceuticals is developing the cell therapy Zimislecel (VX-880), currently in a Phase 1/2 clinical trial. The treatment consists of infusing fully differentiated, stem-cell-derived beta cells into the hepatic portal vein.
Updated clinical data (presented at the ADA conference in June 2024) remains positive:
- 12 patients have achieved insulin independence, some for over two years.
- All participants showed drastic improvements in glycemic control, reaching an HbA1c below the 7% target.
- Adverse events were mostly mild to moderate and related to the required immunosuppressive drugs.
VX-880 is a landmark therapy, being the first to demonstrate that lab-grown cells can restore physiological, glucose-responsive insulin production in humans.
The Evolution of VX-264: A Strategic Pivot
In parallel, Vertex sought to eliminate immunosuppression with VX-264, a program that used the same cells as VX-880 but protected within an encapsulation device.
However, in February 2024, the company announced the discontinuation of this specific study [^1]. The decision was not due to safety but to low efficacy. This “failure” was a crucial engineering lesson, allowing Vertex to direct 100% of its focus toward the most promising strategy: gene-editing the cells themselves to make them “invisible” to the immune system [^2].
⚠️ 3. Current Obstacles
- Ongoing Immunosuppression: The biggest hurdle for VX-880 is the need for anti-rejection medications.
- High Production Cost: Manufacturing clinical-grade (GMP) cells is complex and expensive.
- Access: The path to making this therapy widely available is a long-term challenge.
🔮 4. The Future: Promising, but Selective
Zimislecel (VX-880) is on track to become the first scalable cell therapy for T1D. In the coming years, however, it will likely remain a highly selective treatment. The ultimate goal—a cure without the need for immunosuppression, now being pursued through gene-editing research—is the main focus for Vertex and the entire research field.
Related Readings
- Lantidra: The Therapy That Proved the Concept
- VX-264: The Engineering of the ‘Invisible’ Cell
- The Weapon and the Trigger: Understanding the Origins of T1D
🔗 References
- [^1] Vertex Pharmaceuticals. “Vertex Reports Fourth-Quarter and Full-Year 2023 Financial Results…” Press Release, February 5, 2024.
- [^2] Fierce Biotech. “Vertex axes cell therapy device after early data underwhelm, putting faith in gene-edited prospect.” News, February 6, 2024.
- Data presentation by Vertex Pharmaceuticals at the 84th Scientific Sessions of the American Diabetes Association (ADA), June 2024.
- The New York Times, 2021 and June 20, 2024.