1. Introduction: The Beginning of the End for Diabetes?
For decades, the quest for a cure for Type 1 Diabetes (T1D) resembled trying to fill a leaky bucket. Scientists could create new insulin-producing cells, but the patient’s immune system, the original cause of the disease, would simply destroy the new cells. The only solution was to use immunosuppressive drugs, trading a chronic disease for a permanent state of vulnerability.
In mid-2024, this narrative began to change dramatically. Vertex Pharmaceuticals announced a milestone that seemed to belong to a distant future: the first T1D patient received a dose of beta cells derived from stem cells and edited with CRISPR to become “invisible” to the immune system.
This article reverse-engineers this historic moment. This is not just a new therapy; it is a new paradigm, born from an important lesson in engineering and strategy.
2. The Path to the “Invisible” Cell: A Lesson in Engineering and Strategy
Editor’s Note: The name VX-264 was previously associated with a cell encapsulation program that Vertex discontinued. The company repurposed this designation for its current and more advanced program focused on genetically edited cells. The two technologies are fundamentally different, and their story illustrates a key principle of innovation: learning from mistakes.
The first version of VX-264 attempted to protect the cells with a physical barrier, an encapsulation device. The logic was promising: to avoid immunosuppression. However, the results were not effective enough. In a demonstration of pragmatic management, Vertex discontinued the approach in early 2024, announcing the end of the encapsulation method, but not of the ultimate goal 1. The company redirected its resources to its most advanced technology: gene editing, which was reborn under the same program name.
3. The Engineering of the “Invisible” Cell: Dismantling the Technology
The Holy Grail of regenerative medicine for autoimmune diseases is the creation of a cell that the body accepts without retaliation. Vertex’s approach, consolidated in its VX-264 therapy, attacks the problem on two fronts, using the CRISPR gene-editing tool like a pair of high-precision “molecular scissors.”
- 1. Turning Off the “Intruder Alert” (HLA Editing): All of our cells have proteins on their surface called HLA (Human Leukocyte Antigen). HLA acts as a passport, identifying the cell as “self.” Cells from a donor or a lab-based cell line have a different HLA, which triggers the immune system’s alarm. Vertex uses CRISPR to remove or modify the genes that produce HLA, making the cell essentially anonymous, without a “foreign passport” for inspection.
- 2. Reinforcing the “Don’t Attack Me” Signal (CD47 Overexpression): Removing HLA is not enough. Some immune cells, like macrophages, are programmed to attack any cell that lacks a valid passport. To prevent this, Vertex edits the cells to abundantly produce another surface protein, CD47. This protein functions as a “friendly password,” binding to receptors on immune cells and sending a dominant biochemical signal that means “don’t eat me, I’m one of you.”
This combination of “anonymity” and a “friendly password” is the essence of the hypoimmune cell: a work of biological engineering designed to survive and function in hostile territory.

Concept of the hypoimmune cell: through gene editing, the cell is ‘camouflaged’ to evade detection by the immune system. (Image credit: engeAI.com)
4. The First Human Trial: A Quantum Leap
For years, hypoimmune cell technology has been successfully tested in animal models. However, the announcement that the first human patient has received the VX-264 therapy is an inflection point 2. It is the moment when theory becomes a real clinical possibility. The goal of this first trial (Phase 1/2) is to answer two primary questions:
- Safety: Are the genetically edited cells safe in the human body long-term?
- Proof of Concept: Can the cells indeed survive without immunosuppression and effectively produce insulin?
Initial reports of success, such as the “six months of insulin production” mentioned in the news, are the first sign that the engineering has worked as designed.
5. Vertex’s Strategic Advantage: The Engineering Beyond the Cell
Vertex’s success is not due to an isolated scientific discovery. It is the result of a decade of industrial and engineering strategy that has built a massive competitive advantage.
5.1. The Factory: Mastery of Large-Scale Production (CMC)
While academic labs create cells for research, Vertex has invested hundreds of millions of dollars to turn that science into an industry. With the acquisition of pioneering companies like Semma Therapeutics and ViaCyte, and the construction of state-of-the-art facilities in Boston, Vertex masters CMC (Chemistry, Manufacturing, and Controls). They have developed the process to produce billions of high-quality beta cells consistently and standardized, a feat of bioprocess engineering that no one else possesses at this scale.
5.2. The Ecosystem: Network of Clinics and Accumulated Knowledge
The clinical trial of the previous therapy, VX-880, served as a logistical “dress rehearsal.” With it, Vertex has already built the necessary ecosystem to deliver a cell therapy:
- It established partnerships with the world’s top diabetes and transplant centers.
- It trained teams of doctors, surgeons, and nurses on the infusion and follow-up protocols.
- It developed the cold chain and logistics to transport a living, sensitive biological product from the lab to the patient.
The VX-264 therapy is not starting from scratch. It is being deployed on a “highway” of clinical and logistical infrastructure that has already been built and paved.
5.3. The Hidden Intellectual Property: The Quality Control Patents
Vertex’s true competitive advantage may not just be the edited cell, but the patents on the methods to prove that each batch is safe and effective. Developing the assays that guarantee the absence of genetic errors, the purity of the batch, and its functional potency is an engineering feat in itself, creating an immense barrier to entry for competitors.
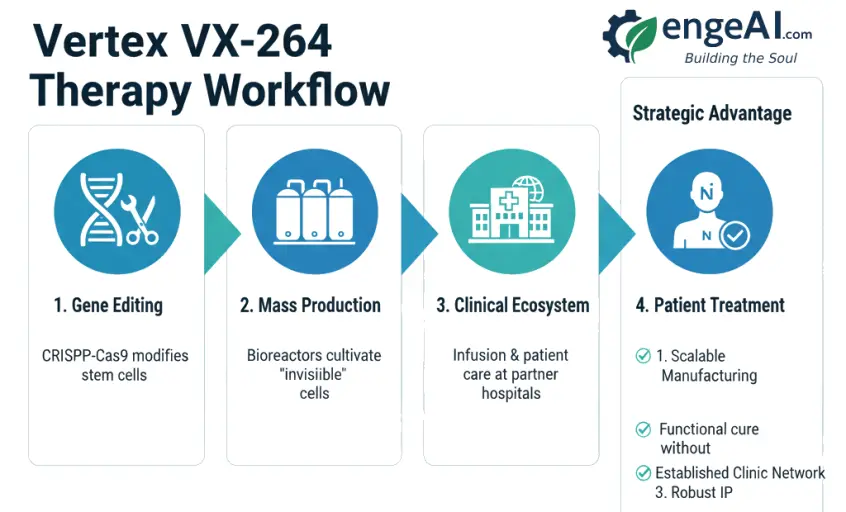
The Vertex ecosystem: from the engineering of the cell to its clinical delivery, illustrating the company’s strategic advantage. (Image credit: engeAI.com)
6. Challenges and Next Steps: The Long Road to the Pharmacy
Despite the optimism, the road is long. Success in one patient needs to be replicated in dozens, and then hundreds. The main challenge is ensuring absolute long-term safety.
- The “Trojan Horse” Risk: A fundamental concern is ensuring the original cell line is 100% free of latent viruses. An “invisible” and contaminated cell could theoretically shield the virus from the immune system, creating a severe safety risk. This requires an unprecedented level of quality control and viral testing.
- Gene-Editing Safety: Regulatory agencies will require years of data to confirm that the CRISPR edits did not cause unintended “off-target effects” in the genome that could manifest in the future.
7. The Engineer’s Conclusions: Realistic Timelines and the Future of Medicine
Vertex’s VX-264 therapy represents the convergence of decades of stem cell biology and the gene-editing revolution. The first human trial marks the beginning of the third era in T1D treatment: the first was insulin; the second, cell therapies with immunosuppression; the third is that of curative regenerative medicine.
- Realistic Timelines: The full path of clinical trials (Phase 1, 2, and 3) and regulatory approval for such a novel therapy is long. Even with positive results, it is unlikely that VX-264 will be commercially available before 2033-2035.
- The Future: The success of this approach will not be limited to diabetes. A platform of “invisible” cells could theoretically be used to treat dozens of other diseases, replacing liver cells, neurons, or heart muscle cells.
For the family living with T1D, the message is one of concrete and unprecedented hope. Engineering is finally beginning to overcome the biology of the disease. The marathon continues, but for the first time, the finish line of a definitive cure is becoming visible on the horizon.
Related Readings
- The Weapon and the Trigger: Understanding the Origins of T1D
- Stopping the Internal War: The Promise of Immunotherapy
- VX-880: The Cell Factory and the Engineering of Scale
- The Personal Bio-Insurance: A Vision for the Future
References
Fierce Biotech. “Vertex axes ViaCyte-partnered diabetes cell therapy program…” News regarding the discontinuation of the encapsulation program, January 2024. ↩︎
Vertex Pharmaceuticals. “Vertex Doses First Patient in Phase 1/2 Clinical Trial of VX-264, a Hypoimmune, CRISPR-Edited Stem Cell Therapy for Type 1 Diabetes.” Press Release (Example). [Search for official press release from Vertex]. ↩︎