1. The Historic Milestone That No One Would Scale
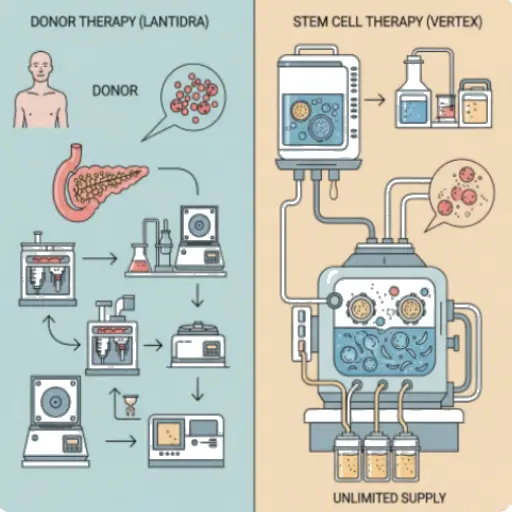
In June 2023, the FDA gave the green light to Lantidra, the first-ever cell therapy for Type 1 Diabetes (T1D). The achievement was undeniable: a procedure that transplants pancreatic islets from deceased donors to patients with the most severe form of the disease proved capable of restoring insulin production and eliminating the need for daily injections 1. For the patient community, it was a ray of light. For industry analysts and investors, however, the approval confirmed a long-held suspicion: the approach, while scientifically valid, is a dead end in terms of scale and access.
Lantidra is an artisanal and exceptional therapy. Its three fundamental barriers are not minor optimizations but intrinsic features of its design:
- The Immunological Barrier: It requires a heavy and continuous regimen of immunosuppression, with all associated risks.
- The Logistical Barrier: The scarcity of pancreas donors is its Achilles’ heel. Sources in the transplant field confirm that, due to the low yield of the isolation process, the pancreases of two to three donors are needed to treat a single patient. This limits the therapy to, at most, a few hundred people per year, in a universe of millions.
- The Economic Barrier: With costs that can exceed $200,000 USD per procedure, Lantidra is intended only for critical health cases, where the risk of severe hypoglycemia outweighs the risks and costs of the treatment 2. It is a niche solution, not a public health one.
2. The Inadvertent Validation: How Lantidra Proved Vertex’s Thesis
Paradoxically, Lantidra’s limited success was the best external validation that Vertex Pharmaceuticals’ strategy could have received. The approval of Lantidra answered the most fundamental question of all for regulators and investors: “does cell replacement therapy for T1D work?”. The answer was a resounding “yes.”
Lantidra proved the biology. With that, it removed the biggest scientific risk from the path, leaving Vertex free to focus on the problem it was born to solve: that of engineering.
The investment thesis in Vertex, which justified the nearly billion-dollar acquisition of Semma Therapeutics, was never about the initial discovery but about the ability to transform a laboratory science into an industrial solution. The company’s strategy rests on two engineering pillars:
Pillar 1: Solving for Scale (The Success of VX-880)
The clinical trial of VX-880 proved the first pillar. Using stem cells, Vertex created an unlimited and standardized source of beta cells, completely eliminating the logistical barrier of donors. Data from conferences like the American Diabetes Association (ADA) show resounding success, with multiple patients becoming insulin-free for over two years 3. VX-880, however, still relied on the same immunosuppression as Lantidra.
Pillar 2: Solving for Immunology (The Pivot to Gene Editing)
The next step was to eliminate the need for immunosuppressants. The first attempt, an encapsulation device (the first version of VX-264), failed due to low efficacy. Instead of insisting, the company made a strategic pivot, learning from the mistake and focusing all resources on the more complex but definitive approach: gene editing (CRISPR) to create a hypoimmune cell.
Industry sources, such as Fierce Biotech, reported this shift not as a failure but as a “demonstration of faith in its gene-editing platform,” the company’s true crown jewel 4.
3. The Cure as an Engineering Problem
Today, the cure for T1D is less a question of “if” and more a question of “how.” Lantidra is the commercial proof-of-concept, but for a minimal market. Vertex is building the industrial solution. Its competitive advantage lies not only in the edited cell but in its ability to mass-produce it with quality (CMC), in its already trained network of hospitals, and, as analysts point out, in the patents on the quality control methods that ensure the safety of each dose.
Lantidra is an excellent, handcrafted concept car for a select few. Vertex is building the gigafactory, the assembly line, and the distribution network for the mass-production model. The approval of the former only made the value of the latter even more evident.
Related Readings
- VX-880: The Cell Factory and the Engineering of Scale
- VX-264: The Engineering of the ‘Invisible’ Cell
- The Weapon and the Trigger: Understanding the Origins of T1D
References
FDA.gov. “FDA Approves First Cellular Therapy to Treat Patients with Type 1 Diabetes.” Press Release, June 28, 2023. ↩︎
Krawiec, R. J., et al. “A systematic review of the cost-effectiveness of islet cell transplantation for the treatment of type 1 diabetes.” Diabetes Technology & Therapeutics, 2020. ↩︎
Data presentation by Vertex Pharmaceuticals at the 84th Scientific Sessions of the American Diabetes Association (ADA), June 2024. ↩︎
Fierce Biotech. “Vertex axes cell therapy device after early data underwhelm, putting faith in gene-edited prospect.” News, February 6, 2024. ↩︎